AUTOCLAVE GUIDELINES / REPAIRS
All autoclave (sterilizers) require annual maintenance to validate compliance and functionality of your equipment. 'Preventative maintenance' can help monitor the condition of a machine over time and greatly improve reliability, but 'invasive maintenance' will always be necessary to restore equipment. The importance of the annual certification is that it satisfies regulatory agency requirements, ensures sterility in the medical field, addresses equipment safety concerns, and offers your staff better training on the equipment in use. Autoclaves come in many forms from: ultrasonic, steam, UV, heat, chemical, and gas. STASIS finds that 'Steam' type sterilizers are the most regularly used machines in the veterinary industry like Tuttnauer, Midmark, and Pelton Cranes. They may also come in 'Automatic' and 'Manual' forms but they will all sterilize the same using steam, pressure, and heat. While a machine may 'work,' without preventive maintenance, your staff can not appropriately validate sterility, safety, or accuracy which can damage the equipment being sterilized and harm the user working with the equipment.
For example, in the pictures above, a reservoir chamber can become extremely dirty, to the extent that the water becomes contaminated before sterilization. When sterilizing, this 'dirty water' will now form into 'steam' but leave behind debris that will bind to your equipment and stain the chamber. This is equivalent to boiling 'waste/sewage' water which will leave behind unsterile equipment and a 'biofilm.' In addition, the remaining sediment from the dirty water, unflushed cleaning solution, pack fiber, and debris will now start to clog pipes, filters, and vent valves. This 'debris' can also block vital sensors from recording input data correctly and prohibit steam from 'venting' and throw up 'error codes.' 'Preventative Maintenance' can replace some of these worn maintenance parts and monitor how much 'clogging' is beginning to accumulate through 'timing scenarios' based on prior years performance. Before pipes and filters begin to constrict and seize closed causing costly repairs and unpredictable downtime, 'invasive maintenance' can clear these lines restoring conditions before they become permanently blocked and obstructed. It is much easier to replace cheap maintenance parts versus expensive pipes that now need extensive labor to be internally removed. This will return the equipment to a 'like new condition' ready again for years of use at a much more affordable rate. These timing scenarios will help better establish 'what is normal' and how severe the cloging is from year to year. However, waiting for the obstruction from clogging to become a 'problem' will create a serious safety risk for the staff at an unpredictable moment. Not only is it much more costly, you may not even get a chance to repair the unit. This 'clogging' is now so severe, it has no way to release pressure due to the blockage and neglect and in essence you will have created a 'pressure cooker bomb.' If the obstruction does not clear, the pressure will increase so greatly internally because it can not vent, it will rupture sending shrapnel throughout the office as it explodes literally like a bomb. You maybe lucky though and it only catches fire from overheating before exploding. Either way, neither 'preventative maintenance' nor 'invasive maintenance' will fix that kind of damage and you will assume all liability because you 'failed to maintain a medical calibrated device.'
In continuation, 'automatic' sterilizers rely heavily on sensors and switches which become faulty over time giving the appearance of being functional, but are actually recording inaccurately. Only by using physical verification tools like thermometers, pressure gauges, multimeters, and sterility checks can they be certified as calibrated. 'Manuals' are typically more reliable but require significant input from a trained operator which if done incorrectly can present unsterile and damaged equipment, and a general safety concern. Part of the 'preventative maintenance' will help you coach your staff on better ways to maintain your equipment and recognize developing problems than can be easily addressed before becoming an unexpected breakdown. Each machine is unique, has its own parts, and cleaning solution. The 'preventive maintenance' performed by STASIS can validate these functions but it is the in-person training that gives you better security knowing your staff is taking care of your unique machine the way the manufacturer recommends until next year for reevaluation.
The equipment being sterilized must also be done by the manufactures recommendations or it will become damaged. Failing to perform preventive maintenance will not only shorten the machines life but it will destroy costly dental handpieces, dull blades, and cause hinges in forceps to seize. The unclean water being used can also create a 'biofilm' that builds up over time and binds to the equipment being inserted causing 'high-speed drills' and 'scalers' to no longer 'spray' water. Not only will it begin to bind to the equipment, but it will also bind to sensors in the chamber like thermostats and pressure gauges and cause errors to arise like 'low temperature,' 'low water,' 'low pressure,' etc.. Especially on 'automatic' units as the 'debris' has now blocked the sensor, the machine creates a continual loop until the sensors are triggered 'on/off.' As it continually goes through the 'action' to trigger the 'sensor,' for example, an 'automatic' machine may try 'fill' a chamber, and will continually fill it with water until it floods because the 'sensor' is 'broken' and it is never told to 'turn off' because the sensor itself is faulty. 'Low Temperature' warnings may occur because it is so dirty by the sensor that the 'biofilm' has actually blocked heat penetration. The sensor will now tell the machine to maximize its temperature output to trigger the 'sensor error' to turn off creating a difference between what is being 'sensed' versus the actually 'internal' temperature. Until the sensor is either cleaned or replaced, the machine will work on 'over-drive' creating more heat than what it is 'sensing' and cause the machine to stress and 'burn-out' the heating element and possibly cause the circuit board to short and even melt. All while damaging any equipment you have placed inside for sterilization. The 'thermometer' used in the maintenance service can validate what 'true' internal temperature is compared to what the gauges and sensors are displaying. Light cleaning may be able to fix the problem but others do require 'invasive maintenance.' Regardless of any maintenance you do, a professional should inspect and validate that the machine is performing correctly in all standards, not just maintenance.
Lastly, but most important are the sterility guidelines for autoclaves that come in two forms: chemical and biological.
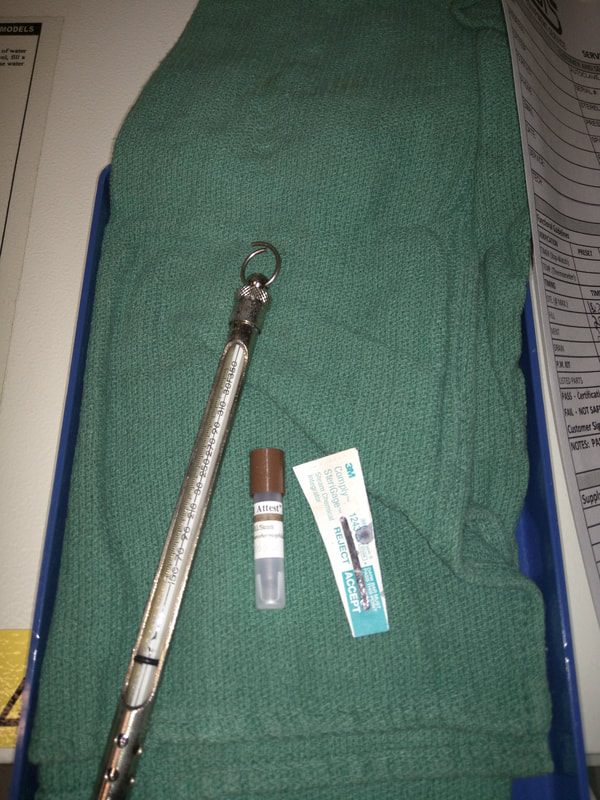
Chemical Integrator (Class 5) Sterility Test - (Class 5 multiparameter) strips are sealed in a 'capsule' filled with both ink and mercury and tested in the autoclave. As pressure begins to rise to a minimum of 15 PSI, the capsule will then begin to 'crack' open allowing the ink and mercury solution to slide up the scale. Mercury will move quicker up the scale based on the internal heat while allowing the ink to track the progress of temperature and time staining the indicator strip. While a high temperature will cause the scale to move, if it does not stay at the correct temperature long enough or looses pressure, your sterility test will fail. This however, gives no confirmation to the 'cleanliness' of the water being used in the sterilizer which leads to the 'Biological Indicator.'
Biological Indicator (Spore) Sterility Test - Uses live 'spores' that have Graham Stained media to detect growth. The sterilizer must penetrate the 'cultured media' inside the 'testing capsule' and literally destroy all microbial life. The 'spores' after a successful 'sterilization' process will not grow after 'incubation' as their DNA has been denatured and no remaining spores will survive in the medical pack to grow back. However, if growth is detected, this is a sign that not only is your machine not 'sterilizing' but potentially an internal 'fungal' growth may exist in your reservoir which means anything inserted into the autoclave is spreading infections materials. 'Invasive Maintenance' will be required at this point to confirm not only function but ensure sterility.
The main 'Certification' of a functioning autoclave with preventative maintenance includes: maintenance procedures, system functions, and sterility validation from Chemical Integrators and Biological Indicators. If you have any questions regarding any of these processes for certification, please 'Contact Us' promptly so we can identify these problems before they get worse.
For example, in the pictures above, a reservoir chamber can become extremely dirty, to the extent that the water becomes contaminated before sterilization. When sterilizing, this 'dirty water' will now form into 'steam' but leave behind debris that will bind to your equipment and stain the chamber. This is equivalent to boiling 'waste/sewage' water which will leave behind unsterile equipment and a 'biofilm.' In addition, the remaining sediment from the dirty water, unflushed cleaning solution, pack fiber, and debris will now start to clog pipes, filters, and vent valves. This 'debris' can also block vital sensors from recording input data correctly and prohibit steam from 'venting' and throw up 'error codes.' 'Preventative Maintenance' can replace some of these worn maintenance parts and monitor how much 'clogging' is beginning to accumulate through 'timing scenarios' based on prior years performance. Before pipes and filters begin to constrict and seize closed causing costly repairs and unpredictable downtime, 'invasive maintenance' can clear these lines restoring conditions before they become permanently blocked and obstructed. It is much easier to replace cheap maintenance parts versus expensive pipes that now need extensive labor to be internally removed. This will return the equipment to a 'like new condition' ready again for years of use at a much more affordable rate. These timing scenarios will help better establish 'what is normal' and how severe the cloging is from year to year. However, waiting for the obstruction from clogging to become a 'problem' will create a serious safety risk for the staff at an unpredictable moment. Not only is it much more costly, you may not even get a chance to repair the unit. This 'clogging' is now so severe, it has no way to release pressure due to the blockage and neglect and in essence you will have created a 'pressure cooker bomb.' If the obstruction does not clear, the pressure will increase so greatly internally because it can not vent, it will rupture sending shrapnel throughout the office as it explodes literally like a bomb. You maybe lucky though and it only catches fire from overheating before exploding. Either way, neither 'preventative maintenance' nor 'invasive maintenance' will fix that kind of damage and you will assume all liability because you 'failed to maintain a medical calibrated device.'
In continuation, 'automatic' sterilizers rely heavily on sensors and switches which become faulty over time giving the appearance of being functional, but are actually recording inaccurately. Only by using physical verification tools like thermometers, pressure gauges, multimeters, and sterility checks can they be certified as calibrated. 'Manuals' are typically more reliable but require significant input from a trained operator which if done incorrectly can present unsterile and damaged equipment, and a general safety concern. Part of the 'preventative maintenance' will help you coach your staff on better ways to maintain your equipment and recognize developing problems than can be easily addressed before becoming an unexpected breakdown. Each machine is unique, has its own parts, and cleaning solution. The 'preventive maintenance' performed by STASIS can validate these functions but it is the in-person training that gives you better security knowing your staff is taking care of your unique machine the way the manufacturer recommends until next year for reevaluation.
The equipment being sterilized must also be done by the manufactures recommendations or it will become damaged. Failing to perform preventive maintenance will not only shorten the machines life but it will destroy costly dental handpieces, dull blades, and cause hinges in forceps to seize. The unclean water being used can also create a 'biofilm' that builds up over time and binds to the equipment being inserted causing 'high-speed drills' and 'scalers' to no longer 'spray' water. Not only will it begin to bind to the equipment, but it will also bind to sensors in the chamber like thermostats and pressure gauges and cause errors to arise like 'low temperature,' 'low water,' 'low pressure,' etc.. Especially on 'automatic' units as the 'debris' has now blocked the sensor, the machine creates a continual loop until the sensors are triggered 'on/off.' As it continually goes through the 'action' to trigger the 'sensor,' for example, an 'automatic' machine may try 'fill' a chamber, and will continually fill it with water until it floods because the 'sensor' is 'broken' and it is never told to 'turn off' because the sensor itself is faulty. 'Low Temperature' warnings may occur because it is so dirty by the sensor that the 'biofilm' has actually blocked heat penetration. The sensor will now tell the machine to maximize its temperature output to trigger the 'sensor error' to turn off creating a difference between what is being 'sensed' versus the actually 'internal' temperature. Until the sensor is either cleaned or replaced, the machine will work on 'over-drive' creating more heat than what it is 'sensing' and cause the machine to stress and 'burn-out' the heating element and possibly cause the circuit board to short and even melt. All while damaging any equipment you have placed inside for sterilization. The 'thermometer' used in the maintenance service can validate what 'true' internal temperature is compared to what the gauges and sensors are displaying. Light cleaning may be able to fix the problem but others do require 'invasive maintenance.' Regardless of any maintenance you do, a professional should inspect and validate that the machine is performing correctly in all standards, not just maintenance.
Lastly, but most important are the sterility guidelines for autoclaves that come in two forms: chemical and biological.
Chemical Integrator (Class 5) Sterility Test - (Class 5 multiparameter) strips are sealed in a 'capsule' filled with both ink and mercury and tested in the autoclave. As pressure begins to rise to a minimum of 15 PSI, the capsule will then begin to 'crack' open allowing the ink and mercury solution to slide up the scale. Mercury will move quicker up the scale based on the internal heat while allowing the ink to track the progress of temperature and time staining the indicator strip. While a high temperature will cause the scale to move, if it does not stay at the correct temperature long enough or looses pressure, your sterility test will fail. This however, gives no confirmation to the 'cleanliness' of the water being used in the sterilizer which leads to the 'Biological Indicator.'
Biological Indicator (Spore) Sterility Test - Uses live 'spores' that have Graham Stained media to detect growth. The sterilizer must penetrate the 'cultured media' inside the 'testing capsule' and literally destroy all microbial life. The 'spores' after a successful 'sterilization' process will not grow after 'incubation' as their DNA has been denatured and no remaining spores will survive in the medical pack to grow back. However, if growth is detected, this is a sign that not only is your machine not 'sterilizing' but potentially an internal 'fungal' growth may exist in your reservoir which means anything inserted into the autoclave is spreading infections materials. 'Invasive Maintenance' will be required at this point to confirm not only function but ensure sterility.
The main 'Certification' of a functioning autoclave with preventative maintenance includes: maintenance procedures, system functions, and sterility validation from Chemical Integrators and Biological Indicators. If you have any questions regarding any of these processes for certification, please 'Contact Us' promptly so we can identify these problems before they get worse.